Nutlin
Nutlins are cis-imidazoline analogs which inhibit the interaction between mdm2 and tumor suppressor p53, and which were discovered by screening a chemical library by Vassilev et al. Nutlin-1, nutlin-2, and nutlin-3 were all identified in the same screen;[1] however, Nutlin-3 is the compound most commonly used in anti-cancer studies.[2] Nutlin small molecules occupy p53 binding pocket of MDM2 and effectively disrupt the p53–MDM2 interaction that leads to activation of the p53 pathway in p53 wild-type cells.[3] Inhibiting the interaction between mdm2 and p53 stabilizes p53, and is thought to selectively induce a growth-inhibiting state called senescence in cancer cells. These compounds are therefore thought to work best on tumors that contain normal or "wild-type" p53. Nutlin-3 has been shown to affect the production of p53 within minutes.[4]
 | |
| Names | |
|---|---|
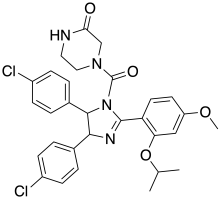
| IUPAC name
(±)-4-[4,5-Bis(4-chlorophenyl)-2-(2-isopropoxy-4-methoxy-phenyl)-4,5-dihydro-imidazole-1-carbonyl]-piperazin-2-one | |
| Other names
Nutlin | |
| Identifiers | |
3D model (JSmol) |
|
| ChEMBL | |
PubChem CID |
|
| UNII | |
| |
| Properties | |
| C30H30Cl2N4O4 | |
| Molar mass | 581.49 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
The more potent of the two enantiomers, (−)-nutlin-3, can be synthesized in a highly enantioselective fashion.[5] Several derivatives of nutlin, such as RG7112 and RG7388 (Idasanutlin) have been developed and progressed into human studies.[6]
References
- Vassilev LT, Vu BT, Graves B, Carvajal D, Podlaski F, Filipovic Z, Kong N, Kammlott U, Lukacs C, Klein C, Fotouhi N, Liu EA (February 2004). "In vivo activation of the p53 pathway by small-molecule antagonists of MDM2". Science. 303 (5659): 844–8. Bibcode:2004Sci...303..844V. doi:10.1126/science.1092472. PMID 14704432.
- Shangary S, Wang S (2008). "Small-molecule inhibitors of the MDM2-p53 protein-protein interaction to reactivate p53 function: a novel approach for cancer therapy". Annual Review of Pharmacology and Toxicology. 49: 223–41. doi:10.1146/annurev.pharmtox.48.113006.094723. PMC 2676449. PMID 18834305.
- Tovar C, Rosinski J, Filipovic Z, Higgins B, Kolinsky K, Hilton H, Zhao X, Vu BT, Qing W, Packman K, Myklebost O, Heimbrook DC, Vassilev LT (February 2006). "Small-molecule MDM2 antagonists reveal aberrant p53 signaling in cancer: implications for therapy". Proceedings of the National Academy of Sciences of the United States of America. 103 (6): 1888–93. doi:10.1073/pnas.0507493103. PMC 1413632. PMID 16443686.
- van Leeuwen IM, Higgins M, Campbell J, Brown CJ, McCarthy AR, Pirrie L, Westwood NJ, Laín S (May 2011). "Mechanism-specific signatures for small-molecule p53 activators". Cell Cycle. Landes Bioscience. 10 (10): 1590–8. doi:10.4161/cc.10.10.15519. PMID 21490429.
- Davis TA, Johnston JN (January 2011). "Catalytic, Enantioselective Synthesis of Stilbene cis-Diamines: A Concise Preparation of (-)-Nutlin-3, a Potent p53/MDM2 Inhibitor". Chemical Science. 2 (6): 1076–1079. doi:10.1039/C1SC00061F. PMC 3375951. PMID 22708054.
- Skalniak L, et al. (November 2018). "Prolonged Idasanutlin (RG7388) Treatment Leads to the Generation of p53-Mutated Cells". Cancers (Basel). 10 (11): 396. doi:10.3390/cancers10110396. PMC 6266412. PMID 30352966.